脾切除对内毒素诱发肝脏脂多糖结合蛋白和TNFα基因表达的影响
杨英祥,陆家齐,杨兴东,冀振华,邱宝安,张志成
陆家齐,杨兴东,冀振华,邱宝安,张志成,中国人民解放军海军总医院肝胆外科 北京市 100037
杨英祥,男,1968-09-27生,安徽省青阳县人,汉族,1993年第二军医大学毕业,1999年军医进修学院外科硕士,肝胆外科主治医师.
项目负责人 杨英祥,100037,北京阜成路6号,中国人民解放军海军总医院肝胆外科. zhhji@95777.com
电话: 010-66958512 传真:010-68574422
收稿日期 2002-03-05 接受日期 2002-03-23
Effect of splenectomy on endotoxin-mediated lipopolysaccharide-binding protein and TNF-α gene expression
Ying-Xiang Yang, Jia-Qi Lu, Xing-Dong Yang, Zhen-Hua Ji, Bao-An Qiu, Zhi-Cheng Zhang
Ying-Xiang Yang, Jia-Qi Lu, Xing-Dong Yang, Zhen-Hua Ji, Bao-An Qiu, Zhi-Cheng Zhang, Department of Hepatobiliary Surgery, Chinese PLA Navy General Hospital, Beijing 100037, China.
Correspondence to: Ying-Xiang Yang, Department of HepatobiliarySurgery, Chinese PLA Navy General Hospital, Beijing 100037, China. zhhji@95777.com
Received 2002-03-05 Accepted 2002-03-23
Abstract
AIM:To investigate the role of lipopolysaccharide-binding protein (LBP) gene expression in the pathogenesis of endotoxemia-mediated systemic inflammation and organ damages after splenectomy.
METHODS:Total 112 male Wistar rats were randomly divided into two groups: control group (n=56), including omentectomy and mobilization of the spleen; s plenectomy group (n=56): including 10 minutes, 0.5, 1.5, 4, 12 and 24 hours subgroups after endotoxin injection. Tissue specimens from liver were obtained. Endotoxin concentrations in the tissue were measured with chromogenic limulus amebocyte lysate (LAL), which was modified by perchloric acid (PCA) pretreatment for samples. LBP and TNF-α mRNA expression were semi-quantitated by the reverse transcription polymerase chain reaction (RT-PCR) using (-actin as an internal standard. Enzyme-linked immunosorbent assay (ELISA) was performed for tumor necrosis factor-α(TNF-α) measurement. The biopsy were carried out for the histological examination.
RESULTS:After endotoxin administration,liver endotoxin concentrations were higher in splenectomized rats than those in controls at 10 minutes(21.5±4.3EU/g vs 10.9±3.3EU/g,P<0.01). Maximal liver LBP mRNA and TNF-α mRNA expression was higher in animals with asplenia than those with eusplenia (LBP mRNA:1.45±0.31 vs 1.09±0.17 TNF mRNA 1.39±0.19 vs 0.97±0.16, P<0.05). Liver TNF-α levels increased markedly in splenectomy groups following endotoxin administration. There was a positive correlation between tissue TNF-αlevels and TNF-α mRNA expression. Similar result also obtained between TNF-α mRNA and LBP mRNA expression. H-E staining showed hepatic cell vacuolation in control groups and steatosis in model groups.
CONCLUSION:Splenectomy can lead to impairment of tissue clearance of endotoxin in liver. Endotoxin accumulated in the local site may markedly up-regulate LBP mRNA expression, and excessive LBP mRNA expression enhances synthesis and release of inflammatory factor. Thus, LBP increases LPS-induced production of inflammatory cytokines.
Yang YX, Lu JQ, Yang XD, Ji ZH, Qiu BA, Zhang ZC. Effect of splenectomy on endotoxin-mediated lipopolysaccharide-binding protein and TNF-α gene expression. Shijie Huaren Xiaohua Zazhi 2002;10(6):654-658
摘 要
目的:观察脾切除动物受内毒素攻击后肝脏脂多糖结合蛋白及肿瘤坏死因子基因表达的变化,探讨脂多糖结合蛋白在脾切除后感染中的可能作用.
方法:♂Wister大鼠112只,随机分为2组:假手术对照组(n=56只):行大网膜切除;脾切除组(n=56只):行脾切除术,每组根据时间点分为内毒素注射前、注射后1/6,0.5,1.5,4,12,24h组;各组均留取肝组织标本,采用改良过氯酸法预处理组织匀浆液、鲎试剂基质显色法检测组织内毒素水平;采用RT-PCR法检测组织LBP,TNF-αmRNA表达水平,ELISA法测定组织TNF-α含量.对肝脏行常规病理检查.
结果:静脉注射内毒素后,脾切除动物肝组织内毒素较有脾动物明显升高(21.5±4.3EU/g vs 10.9±3.3EU/g,P<0.01).脾切除大鼠内毒素攻击后肝组织LBP mRNA 及TNF-α mRNA表达峰值较对照组明显增高(LBP mRNA:1.45±0.31 vs 1.09±0.17,TNF mRNA: 1.39±0.19 vs 0.97±0.16, P<0.05),且峰值持续时间延长,脾切除动物肝组织TNF水平较有脾动物明显增高,相关分析表明,组织TNF-α水平与TNF-α mRNA表达呈显著正相关,TNF-α mRNA表达与LBP mRNA表达显著相关.HE染色光镜下示有脾动物肝细胞空泡及脂肪变性,无脾大鼠可见肝小叶内散在小灶性坏死.
结论:脾切除后机体对内毒素清除延迟,内毒素在组织积聚,组织细胞LBP mRNA过量表达,增敏内毒素激活细胞,进而诱发炎症因子大量产生和全身炎症反应失控,造成器官明显的损害.
杨英祥,陆家齐,杨兴东,冀振华,邱宝安,张志成.脾切除对内毒素诱发肝脏脂多糖结合蛋白和TNFα基因表达的影响.世界华人消化杂志 2002;10(6): 654-658
0 引言
近年来,脂多糖结合蛋白(lipopolysaccharide-binding protein LBP)发现使人们在内毒素(lipopolysaccharide LPS)识别及激活细胞方面获得了突破性进展,即认识到LBP可能是体内极为重要的内毒素雒粝低常湓雒羟慷瓤商岣000倍以上[1-15],大量研究表明,创伤和脓毒症时血清中LBP水平升高,组织内LBP mRNA表达也增加,局部组织内LBP介导的增敏效应可能在器官功能损害发病过程中有着重要的意义[16-27].脾切除术后凶险性感染(overwhelming postsplenectomy infection, OPSI)是指发生于脾切除后的暴发性细菌感染,起病突然,病程特点为迅速演变为休克、低灌注及多器官功能障碍[28-32].我们拟观察脾切除动物受内毒素攻击后肝脏脂多糖结合蛋白和肿瘤坏死因子基因表达及肝脏的组织学变化,以探讨脂多糖结合蛋白在脾切除后感染中的可能作用.
1 材料和方法
1.1 材料 ♂Wistar大鼠112只,体质量200~250g,随机分为2组:无脾组 (脾切除,56只)和有脾组 (假手术对照,56只).实验前禁食过夜,自由饮水.10g/L 戊巴比妥钠麻醉后.取左侧肋缘下斜切口,行全脾切除术, 假手术对照组行部分大网膜切除术和脾移动.术后继续饲养1wk.每组动物均由尾静脉注射内毒素(E.coli O111 B4) 0.1mg/kg,分别于内毒素攻击后1/6,0.5,1.5,4,12,24h处死动物,留取组织标本.置于10%甲醛液,HE染色,观察普通病理变化.
1.2 方法
1.2.1 肝组织内毒素测定 采用改良过氯酸预处理组织匀浆液,用鲎试剂基质显色法(LAL)测定组织内毒素含量.主要步骤:无菌取肝组织0.1g,加入3倍体积无热原生理盐水,冰浴匀浆.吸取组织匀浆液75μL,加入0.32mol/L PCA 150μL,摇匀加盖后37℃水浴20min.离心(3000r/min,15min,4℃)后,取上清50μL加等量0.18mol/L NaOH,混匀后缓慢滴入鲎试剂50μL,加盖37℃水浴22min.然后加入鲎三肽50μL,37℃水浴3min.加入亚硝酸钠0.5mL中止反应,再依次加入氨基磺酸钠0.5mL和萘乙二胺0.5mL,混匀后在DU-7型紫外可见光分光光度计上于波长545nm处测定吸光度值.
1.2.2 肝肺组织TNF-α蛋白含量 采用ELISA法测定,药盒购自美国Biosource公司,操作方法按试剂盒说明书规定进行.试剂盒灵敏度为4ng/L,检测范围为4-1000ng/L,回收率为94-110%.
1.2.3 肝组织LBP及TNF-α mRNA测定 采用RT-PCR半定量法测定肝组织LBP及TNF拨mRNA表达.用异硫氰酸胍一步提取法提取组织总RNA,采用逆转录酶和随机引物进行逆转录合成模板cDNA.通过PCR循环进行基因扩增.PCR步骤:分别向0.5mL离心管中加入10×buffer 2.5μL MgCl2 (20m mol/L) 2.0μL,dNTPs (5m mol/L) 1.0μL,3'5'(5μmol/L)引物1.0μL,逆转录产物3.0μL,去离子水补至25μL.加入石蜡油50μl,97℃变性5min,冷却至88℃时加Taq酶(1U/管)进行PCR循环.大鼠LBP引物序列:5'-CAA ACT CTG CCA GTC ACA-3',5'-GGA CAT TGG CAC CCA AGT-3'[33].大鼠TNF-α引物序列:5'-AGA ACT CCA GGA GGT GTC TGT G-3',5'-GT GGC AAA TCG GCT GAC GGT GT-3'[34].PCR循环产物凝胶电泳,摄像,冲洗后采用LEICA Q-500IW图像分析处理系统进行分析,以积分光密度比值表示mRNA相对表达量.
统计学处理 各项指标均以平均值±标准差(±s)表示,采用SAS(6.04版)统计软件包、Excel(97版)统计函数,不同资料分别进行t检验、Q检验及相关与回归分析等.
2 结果
2.1 注射内毒素前动物肝脏末见明显异常 内毒素静脉注射后12h,有脾动物肝中央静脉周围见炎细胞附壁及集聚,肝细胞水肿空泡变性,无脾大鼠可见肝窦内枯否氏细胞增生,肝小叶内散在小灶性坏死,其中有中性白细胞及淋巴细胞浸润 (图1).
A:对照组肝脏空泡变性
B:脾切除组见小灶性坏死
大鼠肝组织切片 HE×400
2.2 肝组织内毒素水平 内毒素注射前,脾切除和有脾大鼠肝组织含有少量内毒素,内毒素注射后10min,两组动物肝组织内毒素水平均迅速增高,脾切除动物肝组织内毒素升高更明显(21.5±4.3EU/g),与有脾动物(10.9±3.3EU/g)相比差异显著(P<0.01).肝组织内毒素4h后恢复至正常水平(表1).
表1 内毒素攻击大鼠后肝组织内毒素和TNFα水平(x±s)

aP<0.01, bP<0.05, vs 注射前,cP<0.01,dP<0.05, vs 有脾组.
2.3 肝组织LBP mRNA表达 正常肝组织有少量LBP mRNA 表达,内毒素攻击后肝组织LBP mRNA表达迅速增多,1.5h达峰值,脾切除大鼠峰值显著高于有脾大鼠(P<0.05),且峰值持续时间长,4h两组动物仍有明显差异(P<0.05),12h后恢复至伤前基础值(表2).
表2 内毒素攻击后肝TNF-α及LBP mRNA表达(积分A比)
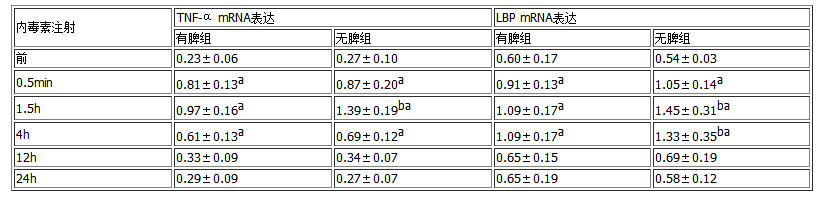
aP<0.01, vs 注射前,bP<0.05, vs有脾组.
2.4 肝组织TNF-α mRNA表达 正常肝组织有少量TNF-α mRNA表达;内毒素攻击后0.5h,脾切除大鼠肝组织TNF-α mRNA表达迅速升高,1.5h达注射前的5倍,峰值更高(增加42.9%倍),与有脾组动物有显著差异(玃<0.05),12h恢复至正常水平(表2).
2.5 肝组织TNF-α水平 内毒素攻击后0.5h肝组织TNF-α水平显著升高,脾切除动物和有脾动物分别升高至注射前的2.62倍和2.25倍,1.5h肝组织TNF-α水平达峰值,脾切除大鼠峰值是有脾大鼠的1.34倍,两组差异显著(P<0.01,表1).肝组织TNF-α浓度与TNF-α,LBP mRNA表达均显著相关(r=0.8547和0.7222,P<0.01),肝组织TNF-α mRNA表达与LBP mRNA表达有明显相关性(r=0.6715,P<0.01).
3 讨论
本研究显示内毒素攻击后10min脾切除动物肝组织内毒素水平明显增高,表明脾切除后组织内毒素的排泄减缓,组织内滞留量增加,提示肝脏在脾切除后器官损害的发生、发展中可能起着重要的调控作用[35-42].LBP在介导细胞应答反应中起显著的增敏作用,临床OPSI病和特点为起病突然,凶猛病情迅速恶化,短期内陷入休克,一般认为革兰氏阴性菌是OPSI的重要病原菌之一[29-31],但LBP增敏系统是否参与脾切除后感染的过程,本实验结果显示,正常肝组织中存在少量LBP mRNA表达,小剂量内毒素(100ng/kg)静脉注射后,无脾大鼠肝组织LBP mRNA表达即迅速增高,1.5h达峰值水平,4h后逐渐下降,12h恢复至正常范围,无脾动物肝组织LBP mRNA表达峰值比有脾大鼠明显增高,1.5h和4h时均存在显著差异,表明脾切除动物感染时器官存在LBP过量表达.LBP具有“酶”的催化作用,可促进内毒素聚体形成单体和加速内毒素与CD14结合, 从而增强细胞对内毒素的敏感性.LBP基因缺陷动物在给予内毒素刺激时不出现急性炎症反应,体内TNF-α水平不增高.大量研究表明, 创伤和脓毒症时血清中LBP水平升高, 组织内LBP mRNA表达也持续增加, 局部组织内LBP介导的增敏效应可能在器官功能损害发病过程中有着重要的意义.
RT-PCR结果表明,无脾大鼠内毒素攻击后肝组织TNF-α mRNA大量表达,说明脾切除可能造成动物免疫功能紊乱,使机体处于一种“预激状态”,同时组织内毒素积聚及LBP mRNA表达增加,促使组织TNF-α mRNA大量表达,TNF-α合成和释放增多.我们即往研究表明,脾切除后较大剂量内毒素(10mg/kg)攻击可造成动物死亡率明显增高,血清中TNF-α水平上升;而给予TNF-α单克隆抗体进行干预,血中TNF-α水平明显降低且死亡率下降,可见TNF-α在无脾动物脓毒症中起着重要的作用[43-45].本实验结果显示,脾切除动物遭受低剂量内毒素(100ng/kg)攻击后30min,肝组织TNF-α水平升高,1.5h达峰值;而有脾大鼠注射内毒素后峰值比无脾大鼠明显降低.同时相关分析表明,肝组织内TNF-α mRNA表达与组织TNF-α浓度显著正相关(r=0.8547, P<0.01),提示TNF-α的调节可能发生在转录水平,调节TNF-α基因转录或翻译可能更有效地阻止脾切除后细菌内毒素的再次打击.本结果还显示,组织TNF-α浓度增高及TNF-α mRNA表达上调与LBP mRNA表达规律相似,相关分析进一步表明,肝组织LBP mRNA与TNF-α mRNA表达具有显著相关性,提示无脾动物内毒素血症时,体内内毒素与LBP形成复合体,广泛激活不同组织合成并释放大量炎症因子,最终导致机体多器官损害.病理证实有脾大鼠受内毒素攻击后,肝脏出现脂肪变性和枯否氏细胞增生,有研究认为这种改变为可逆性的损伤,机体对这种可逆性损伤是能够代偿的,但脾切除大鼠在受到相同剂量的内毒素攻击时,则形成肝脏小灶性坏死,成为不可逆性变化.
我们认为脾切除后机体对内毒素清除延迟,致使内毒素在组织积聚,内毒素刺激组织细胞LBP mRNA表达,大量合成与分泌LBP,LBP-内毒素复合体可增敏内毒素激活细胞,进而诱发炎症因子大量产生和全身炎症反应失控,造成器官明显的损害,最终可能发展成MODS.提示LBP增敏效应可能是临床OPSI病程凶险发展的重要机制之一,通过早期阻抑LBP增敏效应,可能会减缓病情的发展,这为脾切除后凶险感染的预防提供新的思路.
4 REFERENCES
1 Lee PT, Holt PG, McWilliam AS. Role of alveolar macrophages in innateimmunity in neonates: evidence for selective
lipopolysaccharide binding protein production by rat neonatal alveolar macrophages. Am J Respir Cell Mol Biol 2000;23:652-661
2 Echtenacher B, Freudenberg MA, Jack RS, Mannel DN. Differences in innate defense mechanisms in endotoxemia and polymicrobial
septic peritonitis. Infect Immun 2001;69:7271-7276
3 Gutsmann T, Muller M, Carroll SF, MacKenzie RC, Wiese A, Seydel U. Dual role of lipopolysaccharide (LPS)-binding protein in
neutralization of LPS and enhancement of LPS-induced activation of mononuclear cells. Infect Immun 2001;69: 6942-6950
4 Le-Roy D, Di-Padova F, Adachi Y, Glauser MP, Calandra T, Heumann D. Critical role of lipopolysaccharide-binding protein and CD14 in
immune responses against gram-negative bacteria. J Immunol 2001;167:2759-2765
5 Gutsmann T, Haberer N, Carroll SF, Seydel U, Wiese A. Interaction between lipopolysaccharide (LPS), LPS-binding protein (LBP),
and planar membranes. Biol Chem 2001;382:425-434
6 Schroedl W, Fuerll B, Reinhold P, Krueger M, Schuett C. A novel acute phase marker in cattle: lipopolysaccharide binding protein
(LBP). J Endotoxin Res 2001;7:49-52
7 Heinrich JM, Bernheiden M, Minigo G, Yang KK, Schutt C, Mannel DN, Jack RS. The essential role of lipopolysaccharide-binding protein
in protection of mice against a peritoneal Salmonella infection involves the rapid induction of an inflammatory response. J Immunol
2001;167:1624-1628
8 Vreugdenhil AC, Snoek AM, van VC, Greve JW, Buurman WA. LPS-binding protein circulates in association with apoB-containing
lipoproteins and enhances endotoxin-LDL/VLDL interaction. J Clin Invest 2001; 107: 225-234
9 Heumann D, Adachi Y, LeRoy D, Ohno N, Yadomae T, Glauser MP, Calandra T. Role of plasma, lipopolysaccharide-binding protein,
and CD14 in response of mouse peritoneal exudate macrophages to endotoxin. Infect Immun 2001; 69: 378-385
10 Klein RD, Su GL, Schmidt C, Aminlari A, Steinstraesser L, Alarcon WH, Zhang HY, Wang SC. Lipopolysaccharide-binding protein
accelerates and augments Escherichia coli phagocytosis by alveolar macrophages. J Surg Res 2000; 94: 159-166
11 Vreugdenhil AC, Snoek AM, Greve JW, Buurman WA. Lipopolysaccharide-binding protein is vectorially secreted and transported by
cultured intestinal epithelial cells and is present in the intestinal mucus of mice. J Immunol 2000; 165: 4561-4566
12 Jeon YJ, Han SB, Ahn KS, Kim HM. Differential activation of murine macrophages by angelan and LPS. Immunopharmacology
2000; 49: 275-284
13 Dentener MA, Vreugdenhil AC, Hoet PH, Vernooy JH, Nieman FH, Heumann D, Janssen YM, Buurman WA, Wouters EF. Production
of the acute-phase protein lipopolysaccharide-binding protein by respiratory type II epithelial cells: implications for local defense to
bacterial endotoxins. Am J Respir Cell Mol Biol 2000; 23: 146-153
14 Vesy CJ, Kitchens RL, Wolfbauer G, Albers JJ, Munford RS. Lipopolysaccharide-binding protein and phospholipid transfer protein
release lipopolysaccharides from gram-negative bacterial membranes. Infect Immun 2000; 68: 2410-2417
15 Mayeux PR.Pathobiology of lipopolysaccharide. J Toxicol Environ Health 1997; 51: 415-435
16 Yao YM, Yu Y, Fang WH, Zhe HX, Shi ZG, Sheng ZY, Schlag G. The significance of lipopolysaccharide-binding protein and
lipopolysac charide receptor CD14 in increasing sensitivity toendotoxin response and multiple organ damage following trauma.
Chin Critical Care Med 1998; 10: 712-718
17 Hou YF, Mao BL, Qian GS, Xv J. Expression of lipopolysaccharide binding protein mRNA in pulmonary tissues of rat with "two-hit"
acute lung injury and the protective effects of anisodamine in this event. Disan Junyi Daxue Xuebao 2001; 23: 824-826
18 Fang WH, Yao YM, SHI ZG, Yu Y, Wu Y, Lu LR. The significance of the expressions of lipopolysaccharide binding prot ein mRNA and
lipopolysaccharide receptor CD14 mRNA in the liver of burned rat. Chin J Burns 2000; 16: 157-160
19 Wiezer MJ, Meijer C, Sietses C, Prins HA, Cuesta MA Beelen RH, Meijer S, vanLeeuwen PA. Bactericidal/permeability-increasing
protein preserves leukocyte functions after major liver resection. Ann Surg 2000; 232: 208-215
20 Fujimoto M, Uemura M, Nakatani Y, Tsujita S, Hoppo K, Tamagawa T, Kitano H, Kikukawa M, Ann T, Ishii Y, Kojima H, Sakurai S ,
Tanaka R, Namisaki T, Noguchi R, Higashino T, Kikuchi E, Nishimura K, Takaya A, Fukui H. Plasma endotoxin and serum cytokine
levels in patients with alcoholic hepatitis: relation to severity of liver disturbance. Alcohol Clin Exp Res 2000; 24: 48S-54S
21 Kimmings AN, vanDeventer SJ, Obertop H, Rauws EA, Huibregtse K, Gouma DJ.Endotoxin, cytokines, and endotoxin binding
proteins in obstructive jaundice and after preoperative biliary drainage. Gut 2000; 46: 725-731
22 Erwin PJ, Lewis H, Dolan S, Tobias PS, Schumann RR, Lamping N, Wisdom GB, Rowlands BJ, Halliday MI. Lipopolysaccharide
binding protein in acute pancreatitis. Crit Care Med 2000; 28: 104-109
23 Zweigner J, Gramm HJ, Singer OC, Wegscheider K, Schumann RR. High concentrations of lipopolysaccharide-binding protein
in serum of patients with severe sepsis or septic shock inhibit the lipopolysaccharide response in human monocytes.
Blood 2001; 98: 3800-3808
24 Sablotzki A, Borgermann J, Baulig W, Friedrich I, Spillner J, Silber RE, Czeslick E. Lipopolysaccharide-binding protein (LBP) and
markers of acute-phase response in patients with multiple organ dysfunction syndrome (MODS) following open heart surgery.
Thorac Cardiovasc Surg 2001; 49: 273-278
25 Haudek SB, Natmessnig BE, Redl H, Schlag G, Hatlen LE, Tobias PS. Isolation, partial characterization, and concentration in
experimental sepsis of baboon lipopolysaccharide-binding protein. J Lab Clin Med 2000; 136: 363-370
26 Kimmings AN, Vandeventer SJ, Rauws EAJ, Huibregtse K, Gouma DJ. Systemic inflammatory response in acute cholangitis and
after subsequent treatment. Eur J Surg 2000; 166: 700-705
27 Schroder NW, Opitz B, Lamping N, Michelsen KS, Zahringer U, Gobel UB, Schumann RR. Involvement of lipopolysaccharide
binding protein, CD14, and Toll-like receptors in the initiation of innate immune responses by Treponema glycolipids.
J Immunol 2000; 165: 2683-2693
28 Yang YX. The progress in the pathogenesis of overwhelming postsplenectomy infection. Mod Surg 1999;5:59-62
29 Werner AM, Solis MM, Vogel R, Southerland SS, Ashley AV, Floyd JC, Brown C, Ashley DW. Improved antibody responses to
delayed pneumococcal vaccination in splenectomized rats. Am Surg 1999; 65: 844-847
30 Brigden ML, Pattullo AL. Prevention and management of overwhelming postsplenectomy infection--an update.
Crit Care Med 1999; 27: 836-842
31 Waghorn DJ. Overwhelming infection in asplenic patients: current best practice preventive measures are not being followed.
J Clin Pathol 2001; 54: 214-218
32 Yang YX, Lu JQ, Yao YM, Jiao HB, Yu Y, Fu J. The effect of splenectomy on circulating endotoxin clearance and tissue distribution
of endotoxin in rats. Chin J Surg 2000; 38: 787-789
33 Su GL, Freeswick PD, Geller DA. Molecular cloning, characterization, and tissue distribution of rat lipopolysaccharide binding protein,
J Immunol 1994; 153:743-752
34 Estler HC, Grewe M, Gaussling R. Rat tumor necrosis factor-α: transcription in rat kupffer cells and in vitro posttranslational
processing based on a PCR derived cDNA. Biol Chem Hoppe Seyler 1992; 373: 271-281
35 Wang SB, Lu JQ, YU JQ, Lu LR, Yu Y, Yiao YM. The relationship between plasma endotoxin levels and systemic inflammatory
response syndrome in portal hypertensive cirrhosis patients after splenectomy. Chin Crit Care Med 1999;11: 538-540
36 Zhang GF, Zhang MA, Cheng YR, Wang L. The potential role of endothelin and nitric oxide in endotoxin-induced gastric mucosal
damage.Shijie Huaren Xiaohua Zhazhi 2000; 8: 24
37 Liu YQ, Li KY. The characteristic of gastric mucosa in patients with cirrhosis. Shijie Huaren Xiaohua Zhazhi 2000; (Suppl 8): 36
38 Li X, Wang YW. Progress in the study on endotoxemia. Shijie Huaren Xiaohua Zhazhi 2000; (Suppl 8): 114
39 Chi XL, Wang BE, Zhang SW, Zhang NN. Alterations of gastrointestina motility and mucosal barrier in shock rat model induced
by endotoxin plus TNF-α.Shijie Huaren Xiaohua Zhazhi 1999;7:510-512
40 Zhao LF, Han DW. Clinical significance of endotoxemia in liver diseases. Shijie Huaren Xiaohua Zhazhi 1999; 7: 391-393
41 Zhou RR, Liang B. The effect of lipopolysaccharide on hepatocytes in rats in vitro. Shijie Huaren Xiaohua Zhazhi 1999;7:424-425
42 Zhou RR, Liang B. The effect of lipopolysaccharide on hepatocytes cultured at various intervals.
Shijie Huaren Xiaohua Zhazhi 1999;7:431-432
43 Lu JQ, Yang YX, Yiao YM, Yuan SC, Jiao HB, Yu Y. The effect of splenectomy on endotoxin induced tumor necrosis factor
mRNA expression in liver and pulmonary tissue.Chin Crit Care Med 2000; 12: 338-340
44 Yang YX, Lu JQ, Yiao YM, Jiao HB, Yu Y, Fu J. Effect of splenectomy on endotoxin mediated tumor necrosis factor and
interleukin-6 in rats. Chin Crit Care Med 1999; 11: 712-714
45 Jiao HB,Lu JQ,Lu LY, Yu JQ, Wang SB, LI JY. The effect of slenectomy and Escherichia coli endotoxin insult on the plasma
endotoxin TNF and NO levels in rats. Chin J Gen Surg, 1997; 6:78-82

